Acute Optic Neuritis
Privosegtor, a novel peptoid small molecule that penetrates blood brain and retinal barrier, has the potential to become the first neuroprotective therapy for optic neuropathies and other neuro-ophthalmic diseases


Acute optic neuritis is a condition characterized by an acute inflammation of the optic nerve that can lead to permanent visual impairment
Optic neuritis is when inflammation in your optic nerve causes pain, vision loss and other symptoms. This condition has strong links to chronic conditions like multiple sclerosis and other autoimmune diseases. Timely diagnosis and treatment may help optic neuritis and limit or delay more severe long-term effects or conditions.1
It is a type of neuropathy (nerve disease) and happens when inflammation affects signals traveling through your optic nerve, which connects your eyes and brain.
The cells that make up your optic nerve have a fatty coating called a myelin sheath. When you have optic neuritis, that sheath deteriorates. The coating is protective. Without it, the nerve cells can’t send signals properly. That’s why various forms of vision loss are common symptoms of this condition.1
To date there is no specific therapy approved for acute optic neuritis and unmet needs remain for therapies that can prevent vision loss after an acute episode of optic neuritis by avoiding nerve cells damage or death.
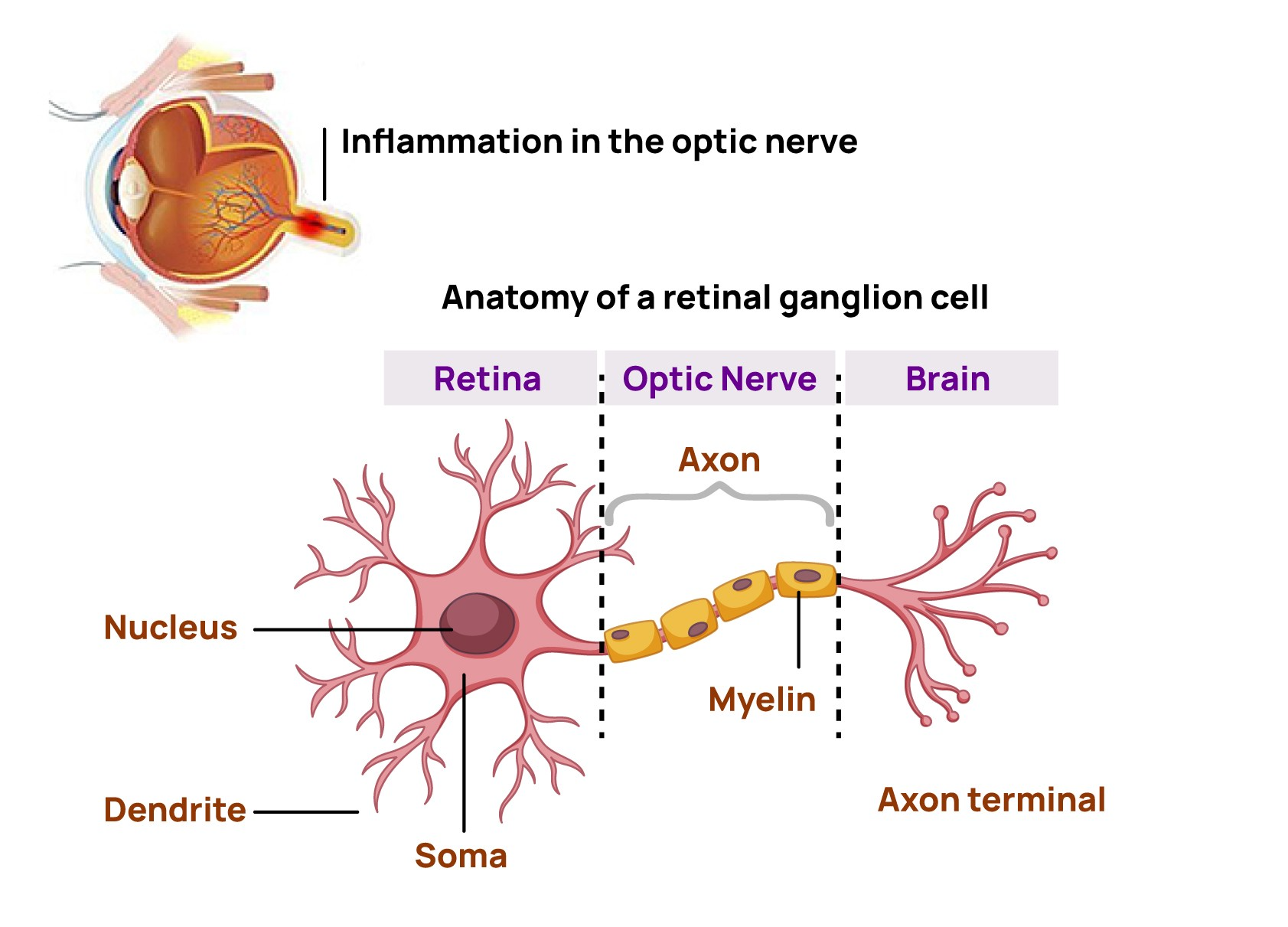
Acute optic neuritis is a condition affecting up to 8 in 100,000 people worldwide2 and often represents the first sign of multiple sclerosis.
It mainly occurs in adults between the age of 20 and 40 years and is more frequent in women (2:1).
Unmet needs remain for neuroprotective therapies that can prevent vision loss after an acute episode of optic neuritis.

Privosegtor
a novel peptoid small molecule that penetrates the blood brain and retinal barrier, with the potential to be a neuroprotective therapy for optic neuropathies and other neuro-ophthalmic diseases.
Privosegtor was selected by high-throughput screening (HTS) for neuroprotective properties, confirmed in vivo in glaucoma, multiple sclerosis, and acute optic neuritis models.
The data from in vitro studies also suggest that it modulates the FOXO3 pathway by interacting with the serum–glucocorticoid kinase (SGK) supporting neuronal survival and preservation.
Privosegtor has shown positive results in the prevention of retinal ganglion cell and axonal damage and was associated with improvements in clinical function (disability) in animal models of neuroinflammation and neurodegeneration.
In 2025, Oculis announced positive results from the ACUITY (Acute OptiC NeUrITis of DemYelinating Origin) trial, evaluating the safety, tolerability and efficacy of OCS-05 in patients with acute optic neuritis.
The trial met the primary endpoint of safety and achieved several key efficacy-based secondary endpoints, showing functional vision improvement and neuroprotective anatomical and biological benefits in patients suffering from acute optic neuritis.
Furthermore, the results of the study show that in addition to the standard of care (IV methylprednisolone), Privosegtor was tolerated in participants with acute optic neuritis. The most frequently reported drug-related treatment-emergent adverse events (TEAEs) in the Privosegtor (2 or 3 mg/kg/day) treatment group were headache and acne, each reported in 2 participants (10.5%).
Privosegtor has received Orphan Drug designation from both the FDA and the EMA for acute optic neuritis.
Privosegtor is an investigational drug and its safety or efficacy has not been established and it has not received regulatory approval for commercial use in any country.
Privosegtor was selected by high-throughput screening (HTS) for neuroprotective properties, confirmed in vivo in glaucoma, multiple sclerosis, and acute optic neuritis models.
The data from in vitro studies also suggest that it modulates the FOXO3 pathway by interacting with the serum–glucocorticoid kinase (SGK) supporting neuronal survival and preservation.
Privosegtor has shown positive results in the prevention of retinal ganglion cell and axonal damage and was associated with improvements in clinical function (disability) in animal models of neuroinflammation and neurodegeneration.
In 2025, Oculis announced positive results from the ACUITY (Acute OptiC NeUrITis of DemYelinating Origin) trial, evaluating the safety, tolerability and efficacy of OCS-05 in patients with acute optic neuritis.
The trial met the primary endpoint of safety and achieved several key efficacy-based secondary endpoints, showing functional vision improvement and neuroprotective anatomical and biological benefits in patients suffering from acute optic neuritis.
Furthermore, the results of the study show that in addition to the standard of care (IV methylprednisolone), Privosegtor was tolerated in participants with acute optic neuritis. The most frequently reported drug-related treatment-emergent adverse events (TEAEs) in the Privosegtor (2 or 3 mg/kg/day) treatment group were headache and acne, each reported in 2 participants (10.5%).
Privosegtor has received Orphan Drug designation from both the FDA and the EMA for acute optic neuritis.
Privosegtor is an investigational drug and its safety or efficacy has not been established and it has not received regulatory approval for commercial use in any country.
Explore Privosegtor Clinical Trial in Acute Optic Neuritis

Phase 2: Privosegtor (OCS-05) in Patients With Acute Optic Neuritis (ACUITY)
Visionary Innovation to Deliver Breakthrough Therapies
Acute optic neuritis is a disease characterized by acute inflammation and demyelination of the optic nerve. While corticosteroids are used to shorten the attack, there is no specific treatment approved for acute optic neuritis, and unmet needs remain for novel therapies that can prevent vision loss.